 
For additional questions and information about buffers see this handout.It would be helpful to model an example first. Differentiation: In lower levels where you do not teach buffers, you could have students do just steps 1-3.You may want to have students write down their answers from the card sort for future reference.If a color printer is not available you can print the cards on different colored card stock, or put the reaction cards (the +, →, NaOH, HCl, and ions) in a separate bag. If possible print the cards in color, since the directions tell them what to do based on the color of the font.I tell them that they can do another reaction if they want until they all are confident with the concept. Groups will finish at different times, so make sure you have something for them to do when they finish.You can use whatever size groups you prefer, but I like having 3-4 in a group so they have more academic conversations. If students are working at lab tables on the card sort, you can walk around and check what they have completed and ask them guiding questions as needed.If one of the cards is found on the floor, you can easily put it back in the right spot. On the back of each card, write that same number. Place each set of cards in a Ziploc bag with a number on the bag.This activity will require a longer amount of teacher preparation the first time (~1 hour) since you will have to cut out the cards.No specific safety precautions need to be observed for this activity.Set of activity cards for each group of students (download file).Teacher Preparation: 1 hour (if cards need to be cut) This activity supports students’ understanding of List two examples of buffers in their everyday life.Explain how a buffer is able to resist changes in pH, regardless of whether an acid or a base is added. 
Write a chemical equation for the reaction of a strong base to a buffer.Write a chemical equation for the reaction of a strong acid to a buffer.Identify a conjugate acid-base pair and determine which one is the acid and which one is the base.īy the end of this activity, students should be able to 
SAP-10.C: Identify the pH of a buffer solution based on the identity and concentrations of the conjugate acid-base pair used to create the buffer.Topic-8.9: Henderson-Hasselbalch Equation. 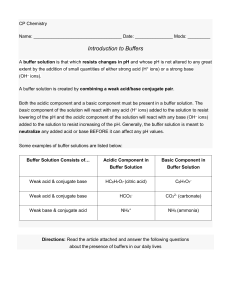
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2022
Categories |
 RSS Feed
RSS Feed
